Strettoi Enrica
 Director of research
Director of research
Via Giuseppe Moruzzi, 1
56124 - Pisa
Tel 050-3153213
Fax 0503153220
This email address is being protected from spambots. You need JavaScript enabled to view it.
Retinal neurobiology in health and disease
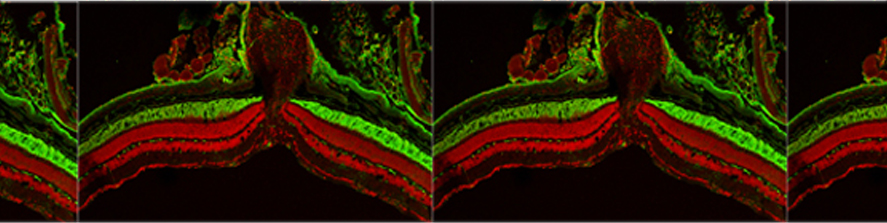
The retina is one of the best-known areas of the Central Nervous System: thanks to modern morphological techniques targeting single neuronal types, the full catalogue of cells (the retinal “neurome”) has been identified for mammalians and the general constitutive plan of this outpost of the brain shown to be highly conserved among Vertebrates. The highly ordered structure of the retina makes it a privileged site for correlations between structure and function so that this organ is studied for its role in vision as well as a paradigm of approachable portion of the CNS to understand its development, physiological properties and response to treatments.
Moreover, retinal diseases often lead to severe visual impairment with high human and economic consequences: this solicits strong investments in basic and applied retinal research worldwide in the attempt to prevent or reduce blindness.
Functional organization of the mammalian retina
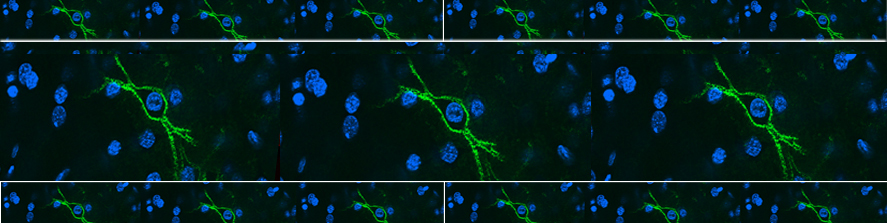
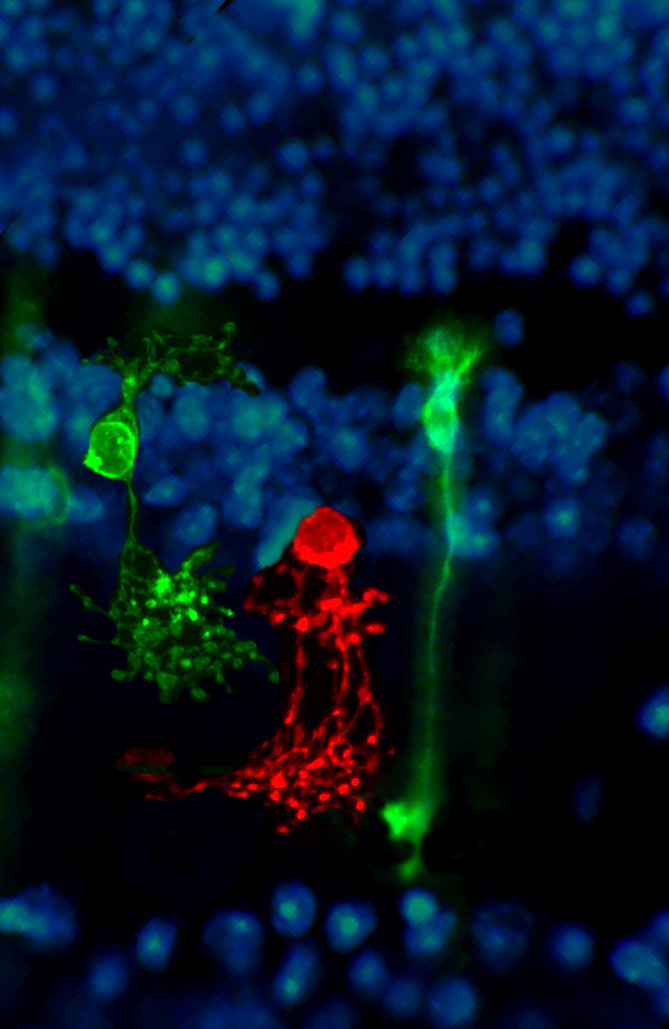
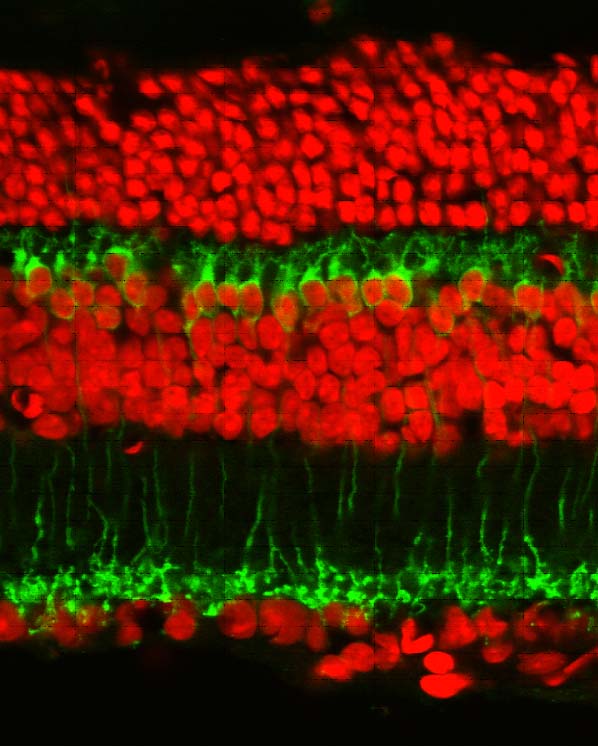
Our research is partly dedicated to the understanding of the functional organization of the normal mammalian retina; we are interested in learning what particular types of neurons are dedicated to specific tasks and what are the consequences of interfering (i.e. by means of genetic manipulations) with their normal development. Examples are studies dedicated to the interactions between rods and cones; to the organization of types of bipolar cells contributing differently to the scotopic and photopic pathways; to the role of ganglion cells in shaping retinal circuitry.
In the years, we have been able to identify general aspects of the anatomical and functional organization of the retina that are highly conserved across different mammalian species and represent a true blueprint.
Neurobiology of inherited photoreceptor degeneration
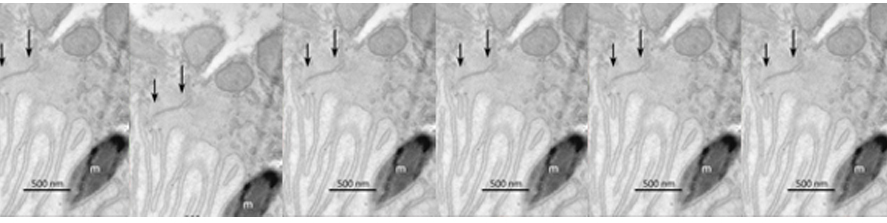
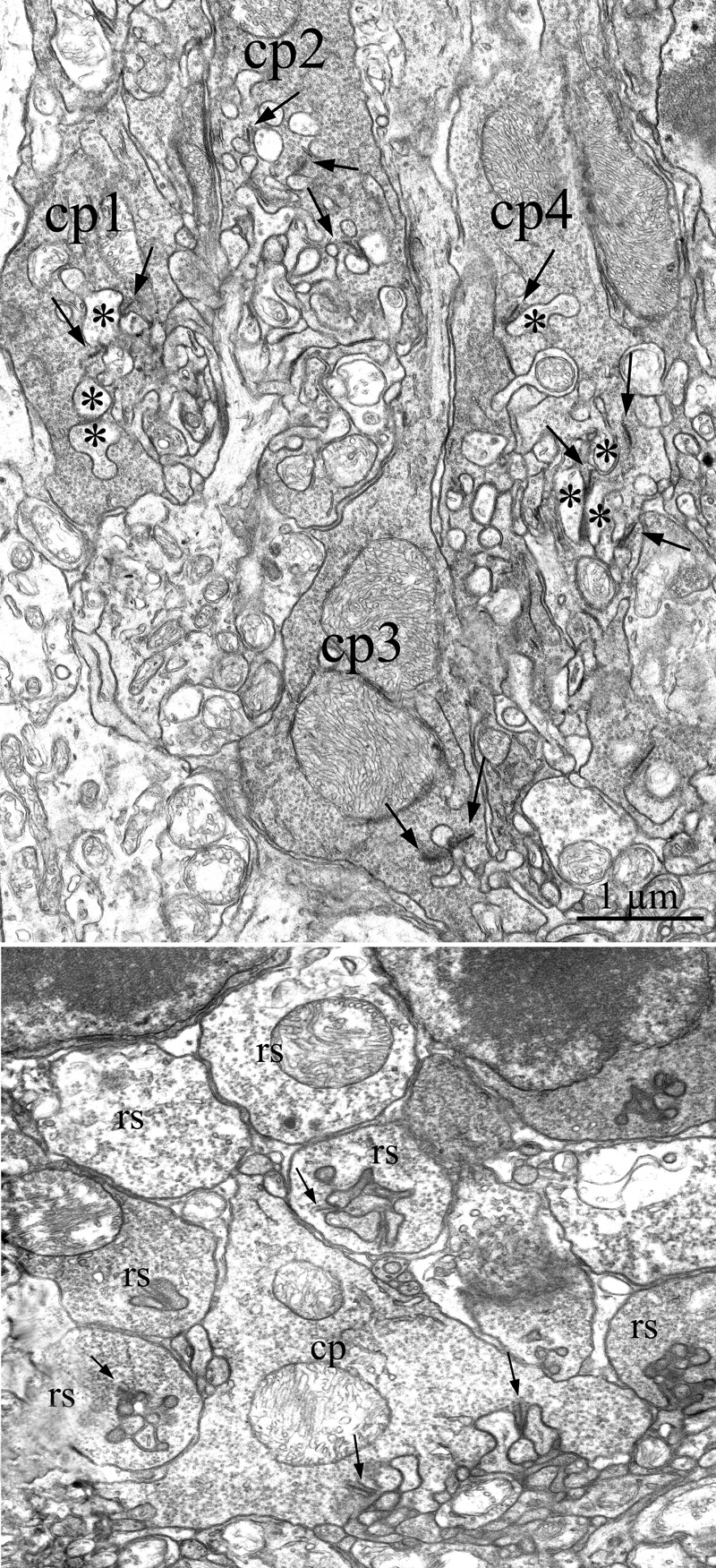
A large fraction of our research is dedicated to exploit animal models of Retinitis Pigmentosa (RP) to understand the pathophysiology of this disease and also to design experimental rescue strategies. RP constitutes a family of inherited disorders causing blindness in approximately 1: 3.500 people. Typically, a mutation (which might be in a photoreceptor-specific gene) leads to the primary degeneration of rods, with consequent night blindness. For still poorly understood reasons, a secondary death of cones occurs, ultimately causing loss of daylight, hi-acuity vision up to legal blindness. Our laboratory has played a relevant role in understanding numerous of the secondary, regressive events slowly propagating to the inner retina, in a process called “remodelling”, which might put to risk repair strategies designed to restore vision.
Because of the dying retinal cells are CNS neurons, and also as a consequence of remodelling and of the high genetic heterogeneity of RP, the possibilities of curing this disease are very limited. However, strategies for retinal repair are actively experimented in many laboratories worldwide and the last decade has experienced tremendous progresses in the field. An important concept is that “sparing the rods, saves the cones”: hence, a simple delay of the primary degeneration of rods can produce a proportionally higher benefit due to a positive effect on cones.
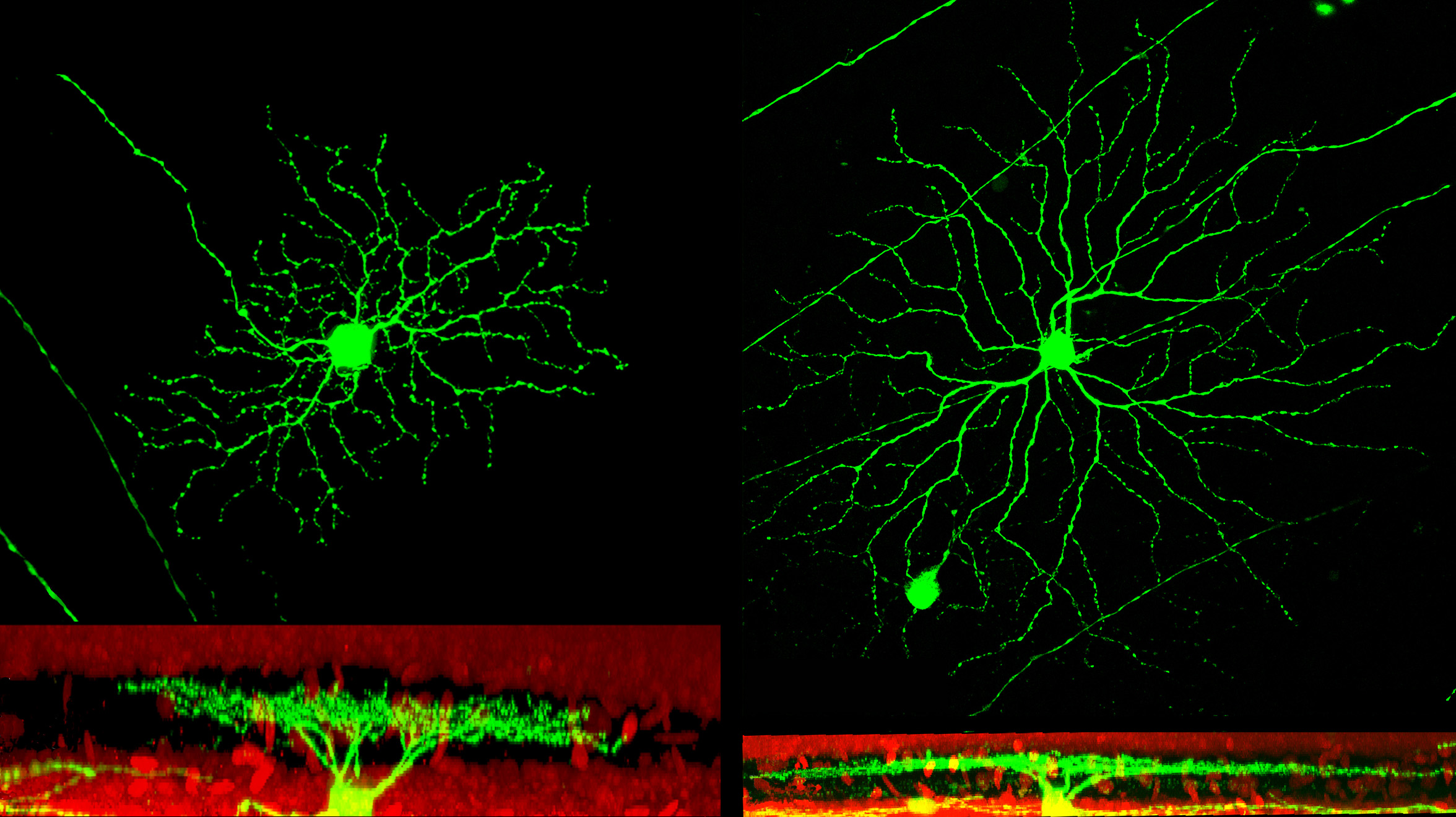
Our effort is to contribute making a cure for RP closer, focussing on these objectives:
- To identify common features of retinal remodelling in paradigms of RP caused by different mutations. The reaction of surviving retinal cells to the death of photoreceptors is crucial, since surviving cells are often the main target of repair therapies, such as optogenetics.
- To exploit rescue therapies based on manipulations of the environment (Environmental Enrichment) to slow down photoreceptor degeneration, enhancing the endogenous defence response of the retina.
- To implement rescue therapies based upon pharmacological tools administered to the eye and targeting selective apoptotic pathways, involved in the primary death of photoreceptors.
Laboratory organization and experimental approach
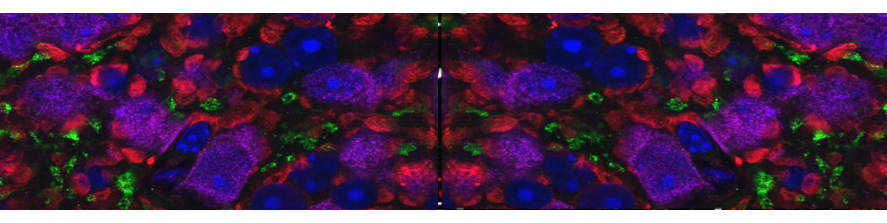
Our research is based on a combination of methods usually beginning with morphological observations and then extend to include biochemical, behavioural, electrophysiological and molecular studies. This multidisciplinary strategy is made possible also by consolidated collaborations with intramural and extramural scientists.
Most recently used approaches comprise: confocal microscopy; image analysis; immunocytochemistry; transmission electron microscopy; immuno-EM; gene-gun dyolistic labelling; western blot; ERG recordings; visual behaviour; qRT-PCR etc.
The laboratory has an international reputation for retinal neurobiology studies and a consolidated network of collaborations with Italian and foreign Institutions.
Representative publications
Strettoi E, Di Marco B, Orsini N, Napoli D. Retinal Plasticity. Int J Mol Sci. 2022 Jan 20;23(3):1138
Puglia C, Santonocito D, Romeo G, Intagliata S, Romano GL, Strettoi E, Novelli E, Ostacolo C, Campiglia P, Sommella EM, Pignatello R, Bucolo C. Lipid Nanoparticles Traverse Non-Corneal Path to Reach the Posterior Eye Segment: In Vivo Evidence. Molecules. 2021 Aug 2;26(15):4673.
Napoli D, Biagioni M, Billeri F, Di Marco B, Orsini N, Novelli E, Strettoi E. Retinal Pigment Epithelium Remodeling in Mouse Models of Retinitis Pigmentosa. Int J Mol Sci. 2021 May 20;22(10):5381.
Thompson DA, Iannaccone A, Ali RR, Arshavsky VY, Audo I, Bainbridge JWB, Besirli CG, Birch DG, Branham KE, Cideciyan AV, Daiger SP, Dalkara D, Duncan JL, Fahim AT, Flannery JG, Gattegna R, Heckenlively JR, Heon E, Jayasundera KT, Khan NW, Klassen H, Leroy BP, Molday RS, Musch DC, Pennesi ME, Petersen-Jones SM, Pierce EA, Rao RC, Reh TA, Sahel JA, Sharon D, Sieving PA, Strettoi E, Yang P, Zacks DN; Monaciano Consortium. Advancing Clinical Trials for Inherited Retinal Diseases: Recommendations from the Second Monaciano Symposium. Transl Vis Sci Technol. 2020 Jun 3;9(7):2.
Piano I, D'Antongiovanni V, Novelli E, Biagioni M, Dei Cas M, Paroni RC, Ghidoni R, Strettoi E, Gargini C. Myriocin Effect on Tvrm4 Retina, an Autosomal Dominant Pattern of Retinitis Pigmentosa. Front Neurosci. 2020 May 6;14:372.
Stefanov A, Novelli E, Strettoi E. Inner retinal preservation in the photoinducible I307N rhodopsin mutant mouse, a model of autosomal dominant retinitis pigmentosa. J Comp Neurol. 2020 Jun 15;528(9):1502-1522.
Testa G, Mainardi M, Morelli C, Olimpico F, Pancrazi L, Petrella C, Severini C, Florio R, Malerba F, Stefanov A, Strettoi E, Brandi R, Arisi I, Heppenstall P,Costa M, Capsoni S, Cattaneo A. The NGF(R100W) Mutation Specifically Impairs Nociception without Affecting Cognitive Performance in a Mouse Model of Hereditary Sensory and Autonomic Neuropathy Type V. J Neurosci. 2019 Dec 4;39(49):9702-9715.
Recent grants (selection)
THE-Tuscany Health Ecosystem. A PNRR project funded by the Italian MUR. 2022-2025
Pharmacological strategies for mutation-independent treatments of Retinitis Pigmentosa. Velux Foundation, Switzerland. 2019-2023
In the eye of the observer: Visual processing at the heart of the retina (Switchboard). European Union Innovative Training Network, Marie Curie Action. 2015-2019
Recent honors
2023- Silver Fellow of ARVO, the Association for research in Vision and Ophthalmology
2021- Nominated among the Top Women in European Vision Research and Ophthalmology, European Vision Institute, EVI
2016 - International Research Collaborative Award IRCA), University of Sydney, Australia. Award to support a period of research in the laboratories of the Save Sight Institute (Sydney) for studies on the primate retina (July-September 2016).
Long-term collaborations
Claudia Gargini, Department of Pharmacy, University of Pisa, Italy
Antonino Cattaneo, Scuola Normale Superiore, Pisa
Simona Capsoni, University of Ferrara
Stanislao Rizzo, Policlinico Gemelli, Rome
Strettoi's biography (09 june 2023)