Kaludercic Nina
- Dettagli
-
Visite: 11667
 Research scientist
Research scientist
c/o Complesso Biologico Interdipartimentale
A. Vallisneri
Viale Giuseppe Colombo 3
35121 Padova
Tel 049-8276414
Fax 049-8276040
Questo indirizzo email è protetto dagli spambots. È necessario abilitare JavaScript per vederlo.
On leave 2023-2025
Currently c/o Dipartimento di Scienze Biomediche, Università degli Studi di Padova
email: Questo indirizzo email è protetto dagli spambots. È necessario abilitare JavaScript per vederlo.
web page: https://www.biomed.unipd.it/category/ruoli/personale-docente?key=0E20B50B5C031AED6BB8FD735D4CBA31
Monoamine oxidase involvement
in oxidative stress and mitochondrial
dysfunction in cardiovascular diseases
Research summary
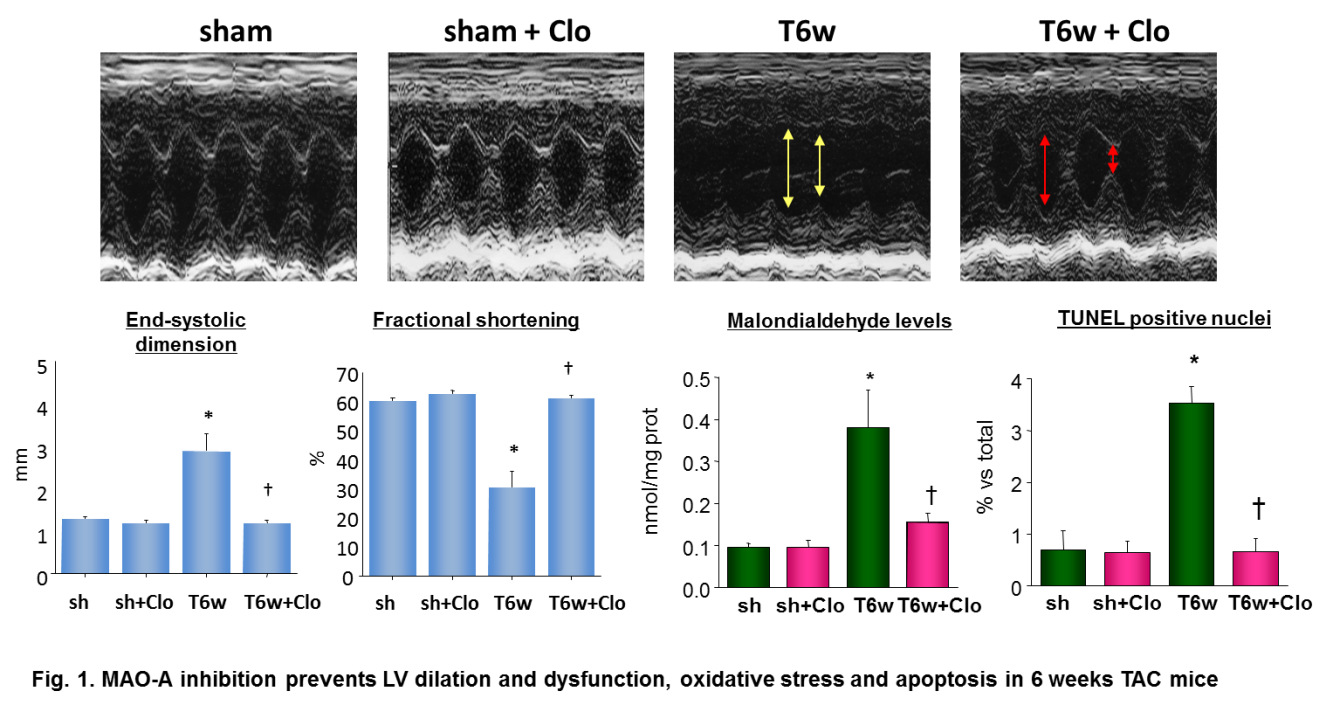
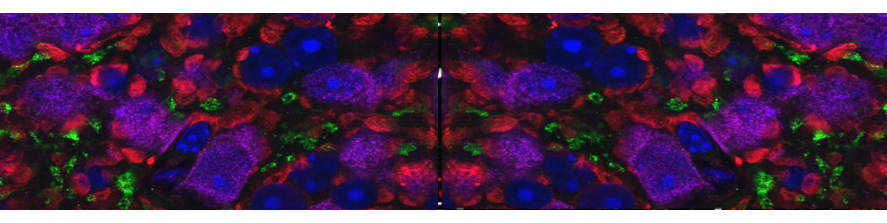
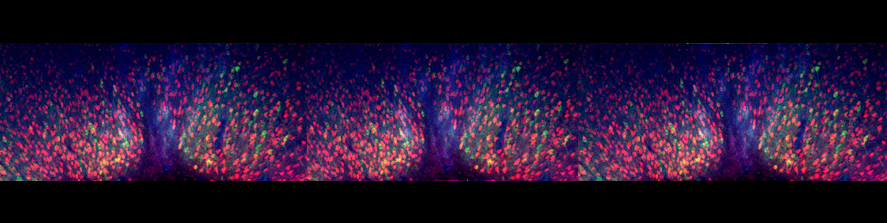
Changes in cardiac tissue redox balance participate in myocyte hypertrophy and failure, influencing ECM remodeling, Ca2+ handling, and metabolic substrate. Identification of the important sources of reactive oxygen species (ROS) in the cell is therefore of major importance. In the cytosol, NADPH oxidase, xanthine oxidase and uncoupled nitric oxide synthase are recognized ROS sources. Mitochondria are another major source, largely from the respiratory chain and p66Shc, and are known to contribute to ischemia/reperfusion injury, diabetes and heart failure. Within the mitochondria, monoamine oxidases (MAO) are a major source of ROS. These flavoenzymes are localized at the level of the outer mitochondrial membrane and exist in two isoforms, MAO-A and –B and are responsible for the catabolism of neurotransmitters and biogenic amines, generating hydrogen peroxide during this process. Previous work from our group showed that MAO-A plays a central role in maladaptive hypertrophy and heart failure. MAO-A is responsible for enhanced norepinephrine catabolism in failing hearts, leading to increased oxidative stress, apoptosis, left ventricle remodeling and dysfunction (Fig. 1). Importantly, both pharmacological and genetic inhibition of this enzyme is sufficient to prevent all these changes.
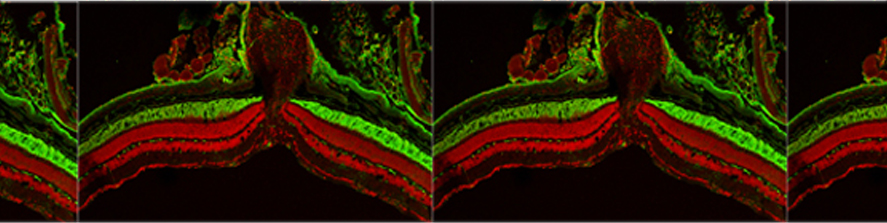
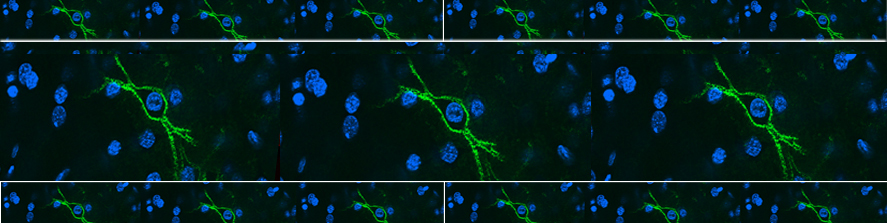
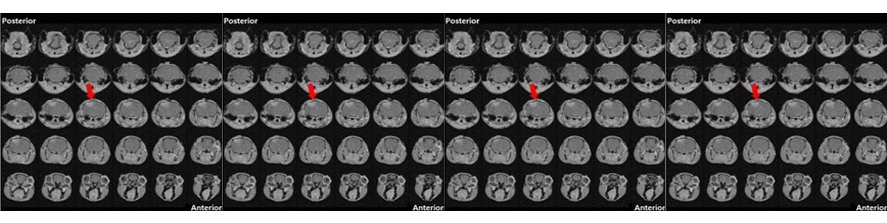
 Recent study published from our group, showed that MAO-B isoform, predominantly expressed in mouse and human heart, also plays a major role in determining the transition of concentric, compensated hypertrophy to heart failure through generation of ROS. Furthermore, we showed that ROS and aldehydes generated through MAO-B-dependent dopamine degradation are able to directly target mitochondria and induce mitochondrial dysfunction (Fig. 2).
Recent study published from our group, showed that MAO-B isoform, predominantly expressed in mouse and human heart, also plays a major role in determining the transition of concentric, compensated hypertrophy to heart failure through generation of ROS. Furthermore, we showed that ROS and aldehydes generated through MAO-B-dependent dopamine degradation are able to directly target mitochondria and induce mitochondrial dysfunction (Fig. 2).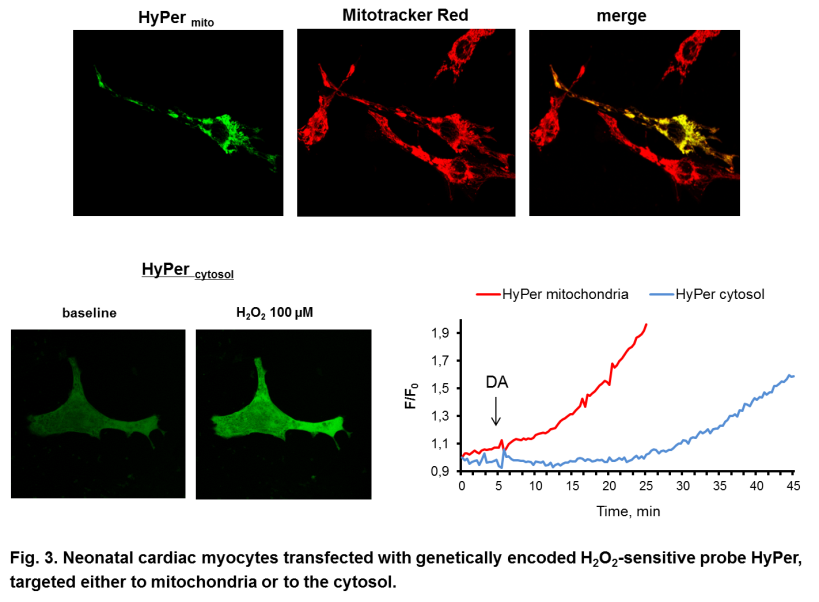
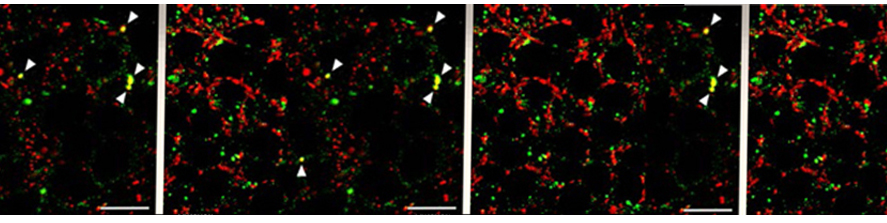
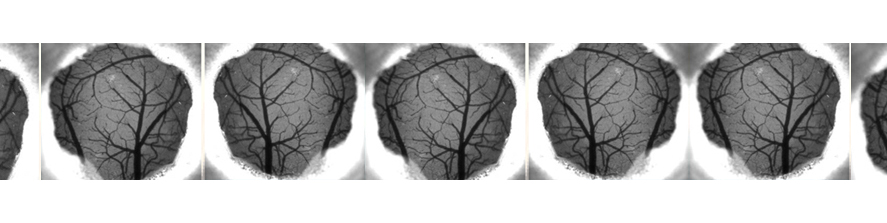
Using genetically encoded redox sensitive probe that can be targeted specifically to mitochondria or to the cytosol, we showed that MAO activation results in H2O2 generation predominantly in the mitochondria that ultimately leads to the loss of mitochondrial membrane potential (Fig. 3).
Currently I am investigating the role of MAOs in diabetic cardiomyopathy, how post-translational changes of MAO protein may impact its activity and how cellular and mitochondrial redox state might regulate protein expression.
Member of group
Cardiac biochemistry
Representative publications
Kaludercic N, Mialet-Perez J, Paolocci N, Parini A, Di Lisa F:
Monoamine oxidases as sources of oxidants in the heart. J Mol Cell Cardiol, in press.
Kaludercic N, Carpi A, Nagayama T, Sivakumaran V, Zhu G, Lai EW, Bedja D, De Mario A, Chen K, Gabrielson KL, Lindsey ML, Pacak K, Takimoto E, Shih JC, Kass DA, Di Lisa F, Paolocci N:
Monoamine Oxidase B Prompts Mitochondrial and Cardiac Dysfunction in Pressure Overloaded Hearts. Antioxid Redox Signal. 2014 Jan 10;20(2):267-80.
Kaludercic N, Takimoto E, Nagayama T, Lai EW, Feng N, Bedja D, Shih JC, Chen K, Gabrielson KL, Pacak K, Blakely RD, Kass DA, Di Lisa F, Paolocci N:
Monoamine oxidase A mediated enhanced catabolism of norepinephrine contributes to adverse remodeling and pump failure in hearts with pressure overload. Circ Res. 2010 Jan 8;106(1):193-202.
Agnetti G, Kaludercic N, Kane L, Elliott ST, Guo Y, Chakir K, Samantapudi D, Paolocci N, Tomaselli GF, Kass DA, Van Eyk JE:
Modulation of mitochondrial proteome and improved mitochondrial function by biventricular pacing of dyssynchronous failing hearts. Circ Cardiovasc Genet. 2010 Feb 1;3(1):78-87.
Kaludercic N, Carpi A, Menabò R, Di Lisa F, Paolocci N:
Monoamine oxidases (MAO) in the pathogenesis of heart failure and ischemia/reperfusion injury. Biochim Biophys Acta. 2010 Sep 24.
 Research scientist
Research scientist

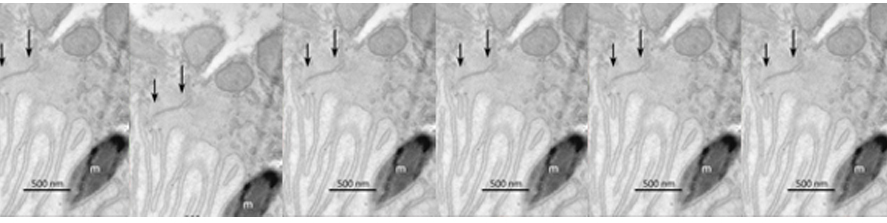

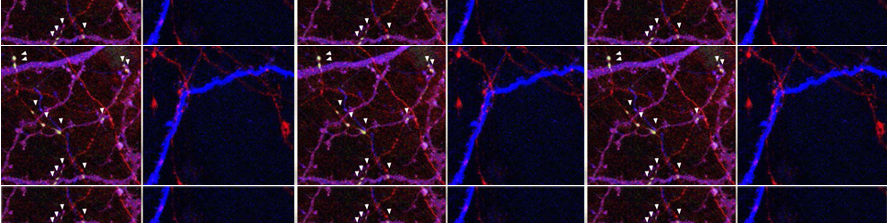

 Recent study published from our group, showed that MAO-B isoform, predominantly expressed in mouse and human heart, also plays a major role in determining the transition of concentric, compensated hypertrophy to heart failure through generation of ROS. Furthermore, we showed that ROS and aldehydes generated through MAO-B-dependent dopamine degradation are able to directly target mitochondria and induce mitochondrial dysfunction (Fig. 2).
Recent study published from our group, showed that MAO-B isoform, predominantly expressed in mouse and human heart, also plays a major role in determining the transition of concentric, compensated hypertrophy to heart failure through generation of ROS. Furthermore, we showed that ROS and aldehydes generated through MAO-B-dependent dopamine degradation are able to directly target mitochondria and induce mitochondrial dysfunction (Fig. 2).