Di Benedetto Giulietta
- Dettagli
-
Visite: 9738
 Research scientist
Research scientist
c/o Complesso Biologico Interdipartimentale
A. Vallisneri
Viale Giuseppe Colombo 3
35121 Padova
Tel 049-7923239
Fax 049-7923250
Questo indirizzo email è protetto dagli spambots. È necessario abilitare JavaScript per vederlo.
Mitochondrial signalling
Research summary
My research interests include cAMP and Ca2+signalling and their subcellular compartmentalization in microdomains; in particular my recent activity has been directed to the study of cAMPsignalling impacting mitochondria, both from the cytosol and from the matrix side.
My main research is presently focused on the role of matrix cAMP in the regulation of mitochondrial bioenergetics.
Role of matrix cAMP in the regulation of mitochondrial bioenergetics
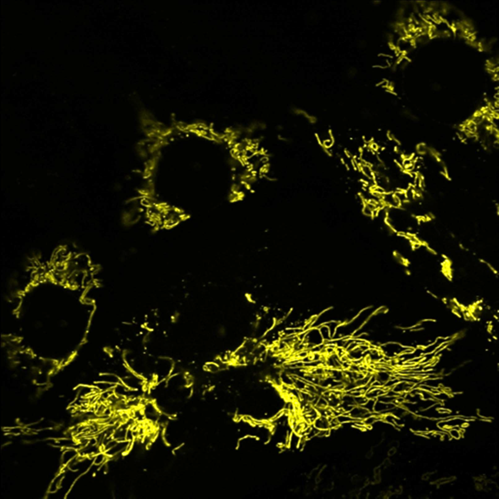
Mitochondria, the cell powerhouse, are dynamic organelles that participate in multiple cellular functions, including apoptosis, autophagy, metabolic control and intracellular signalling. Mitochondria are involved in processes as diverse as development, cancer progression, neurodegeneration and aging, and a tight linkage between mitochondrial dysfunction and major human diseases is increasingly emerging.
Mitochondria constantly respond to changes in substrate availability and energy utilization to maintain cellular ATP supplies; reversible phosphorylation of mitochondrial proteins has been proposed to play a fundamental role in metabolic homeostasis, but little is known about the signaling pathways involved. cAMP is known to play several different roles in distinct cellular compartments but its role on mitochondrial functions has only recently received experimental support, and still remains largely unknown.
To investigate the role of cAMP within mitochondria, we have targeted a FRET-based cAMP biosensor to the mitochondrial matrix of living cells. This, in conjunction with other genetic and pharmacological tools, allowed us to directly show that a mitochondrial form of soluble adenylate cyclase (sAC) is responsible for the generation of cAMP inside the organelles, and that matrix cAMP regulates the organelles ATP synthesizing efficiency. We have also shown that cAMP and Ca2+signalling cross-talk within the matrix, with Ca2+ increases stimulating cAMP production. In cardiac myocytes, mitochondrial cAMP can be increased by adrenergic stimulation, which augments frequency and amplitude of Ca2+ oscillations.
The existence, within mitochondria, of a hitherto unknown crosstalk between cAMP and Ca2+ opens the way to a re-evaluation of these organelles not only as key controller of Ca2+signalling, but also as integrators of multiple second messengers.
We now plan to extend our analysis to define the identity of mitochondrial cAMP effectors and targets, and to elucidate the impact of cAMPsignalling on mitochondrial function
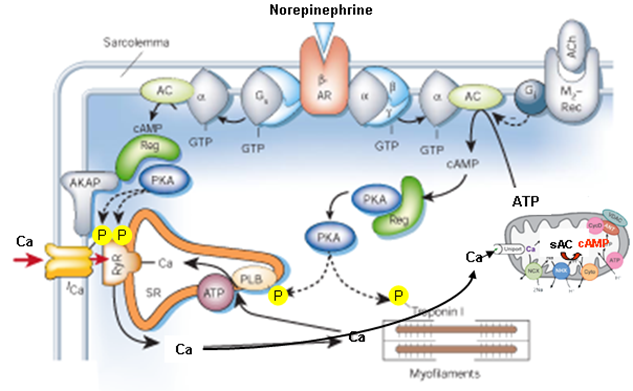
In cardiac myocytes, cAMPgenerated in response to beta-AR stimulation modulates excitation-contraction coupling by activating, via PKA, the phosphorylation of several different targets, thus increasing the amount of Ca2+ available for contraction. The amplified mitochondrial Ca2+cycling activates the generation of mitochondrial cAMP.
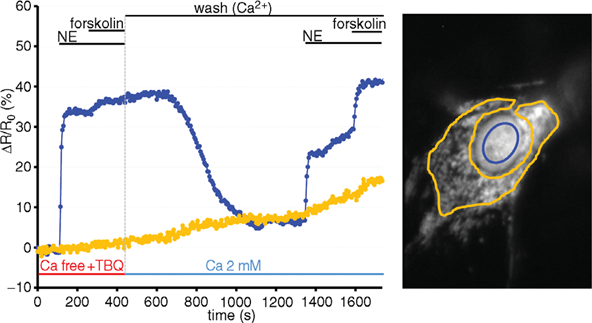 Representative kinetics of cAMP changes recorded in a rat neonatal cardiomyocyte co-expressing a mitochondrial (yellow) and a nuclear (blue) cAMP sensor are presented. The cell was stimulated, in sequence, with norepinephrine (NE) and forskolin. This double stimulation has been performed twice: initially in the absence of Ca2+and then after washing the drugs and in the presence Ca2+in the medium. For details, see Di Benedetto et al. (2013).
Representative kinetics of cAMP changes recorded in a rat neonatal cardiomyocyte co-expressing a mitochondrial (yellow) and a nuclear (blue) cAMP sensor are presented. The cell was stimulated, in sequence, with norepinephrine (NE) and forskolin. This double stimulation has been performed twice: initially in the absence of Ca2+and then after washing the drugs and in the presence Ca2+in the medium. For details, see Di Benedetto et al. (2013).
Representative publications
Di Benedetto G., Scalzotto E., Mongillo M., Pozzan T., Mitochondrial Ca2+ uptake induces cAMP generation in the matrix and modulates organelle ATP levels. Cell Metab 17(6):965-75 (2013)
Di Benedetto G., Pendin D., Greotti E., Pizzo P., Pozzan T., Ca2+ and cAMP crosstalk in mitochondria. J Physiol (2013), Epub ahead of print.
Gomes L.C., Di Benedetto G., Scorrano L., During autophagy mitochondria elongate, are spared from degradation and sustain cell viability. Nat Cell Biol. 13(5):589-98 (2011).
Di Benedetto G., Zoccarato A., Lissandron V., Terrin A., Li X., Houslay M.D., Baillie G., Zaccolo M., Protein Kinase A Type I and Type II Define Distinct Intracellular Signalling Compartments. Circ Res 103 (8): 836-44 (2008)
A. Terrin, G. Di Benedetto, V. Pertegato, Y.-F. Cheung, G. Baillie, M.J. Lynch, N. Elvassore, A. Prinz, F.W. Herberg, M.D. Houslay, and M. Zaccolo., PGE1 stimulation of HEK293 cells generates multiple contiguous domains with different [cAMP]: role of compartmentalized phosphodiesterases. J Cell Biol 175 (3): 441-451 (2006)
Cano Abad M.F.,Di Benedetto G., Magalhaes P.J., Filippin L., Pozzan T. , Mitochondrial pH monitored by a new engineered GFP mutant. J BiolChem 279 (12): 11521-9 (2004).
 Research scientist
Research scientist

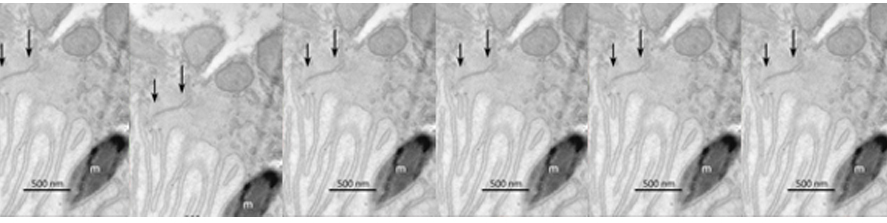

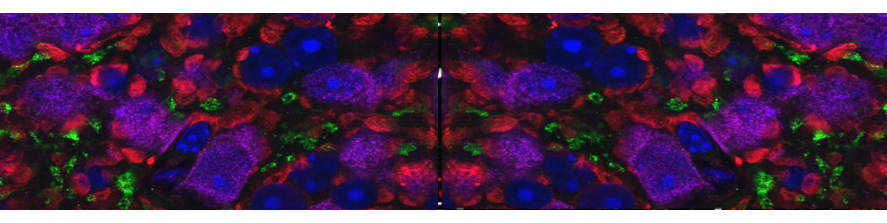
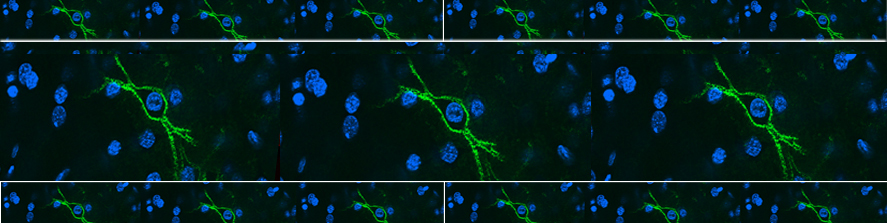
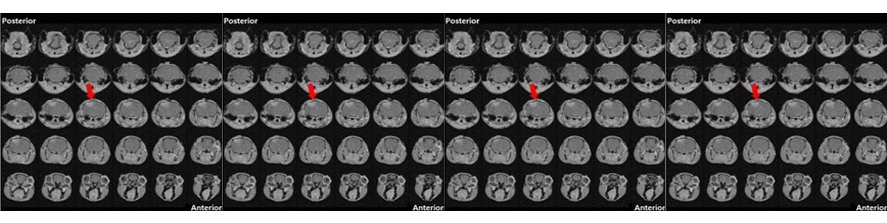
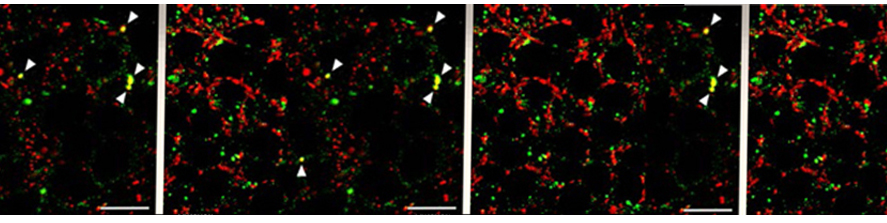
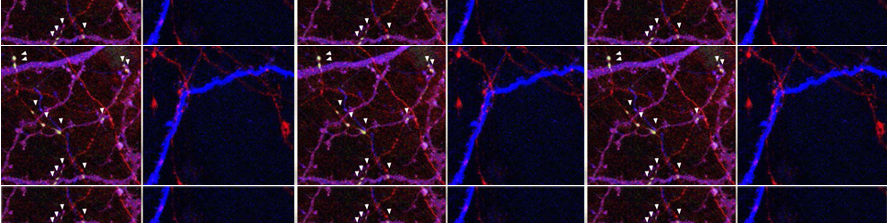


 Representative kinetics of cAMP changes recorded in a rat neonatal cardiomyocyte co-expressing a mitochondrial (yellow) and a nuclear (blue) cAMP sensor are presented. The cell was stimulated, in sequence, with norepinephrine (NE) and forskolin. This double stimulation has been performed twice: initially in the absence of Ca2+and then after washing the drugs and in the presence Ca2+in the medium. For details, see Di Benedetto et al. (2013).
Representative kinetics of cAMP changes recorded in a rat neonatal cardiomyocyte co-expressing a mitochondrial (yellow) and a nuclear (blue) cAMP sensor are presented. The cell was stimulated, in sequence, with norepinephrine (NE) and forskolin. This double stimulation has been performed twice: initially in the absence of Ca2+and then after washing the drugs and in the presence Ca2+in the medium. For details, see Di Benedetto et al. (2013).